✅ This is a DIGITAL DOWNLOAD, no physical product will be shipped.
I made sure to include every possible way I have ever seen on exams that teachers can ask a question about pH, pOH, [H+], [OH-] and everything on Acid Base Equilibria. These guides are exactly what you need to feel confident on your next exam!
A complete how-to guide on:
- Important acid and base definitions
- Strong and weak acids definitions and list
- Strong and weak bases definitions and list
- Acid-base neutralization reactions
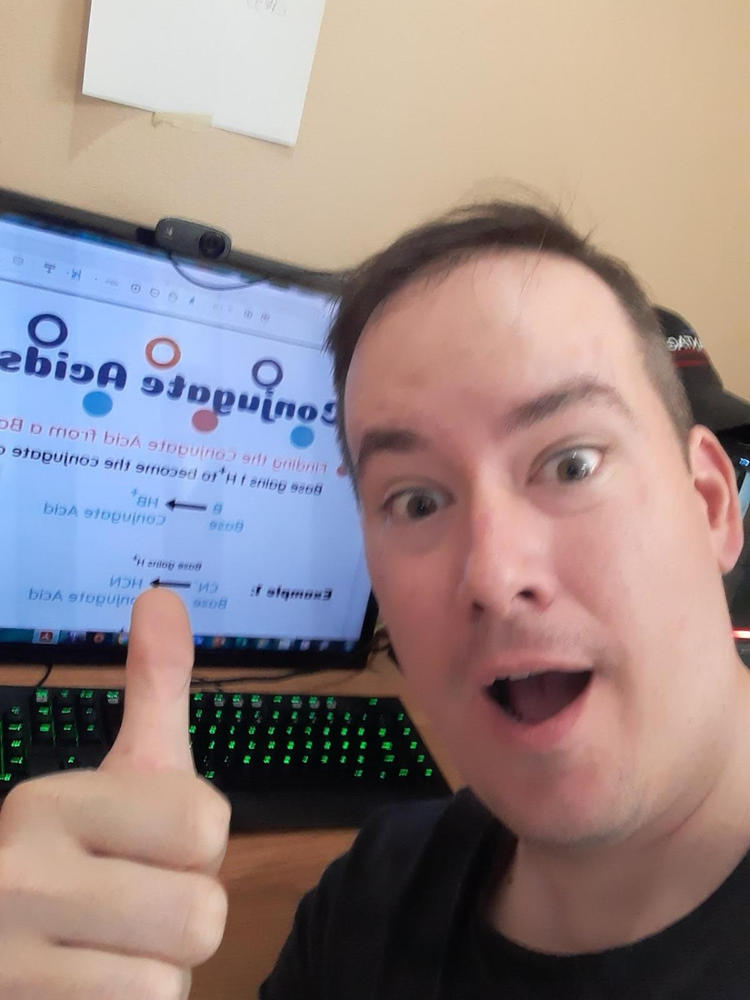
- How to find conjugate acids and bases
- Amphoteric species
- Finding pH, pOH, [H+] and [OH-]
- Using the 5% Rule (x is small assumption)
- Using the Quadratic Formula
- Finding the pH of Strong and Weak Acids
- Finding the pH of Strong and Weak Bases
- Finding Ka and Kb
- Percent Ionization
- Classifying Salts
- Classifying Salts using Dissociation Equations
- Detailed examples that explain every single step
- Conjugate acid and base pairs explained and why charges change
- List of strong acids and weak acids
- List of strong bases and weak bases
- Common multiple choice and test questions
- Tips and tricks
- All of the possibilities you may see when finding pH, pOH, [H+] and [OH-]
After your purchase, you'll receive an email with a download link.
Please note: There are no refunds on digital guides since there is no way to "un-download" them.
Perfection
I’m blessed to have found you on YouTube... I honestly tell everyone about you and my notes got tons of attention in class!!! Melissa Maribel has become a household name in my family !!!
Good study guide
The study guide has everything and it has been broken down so its easier to understand.
Extremely Helpful!
The notes are really pretty and well-organized. It is helpful having links in the Table of Contents to take you to any topic you need help with. This has helped me with Chem 104.
Amazing
Made the homework a thousand times easier to complete, thank you Melissa!
Great
Really helped me learn first year chem